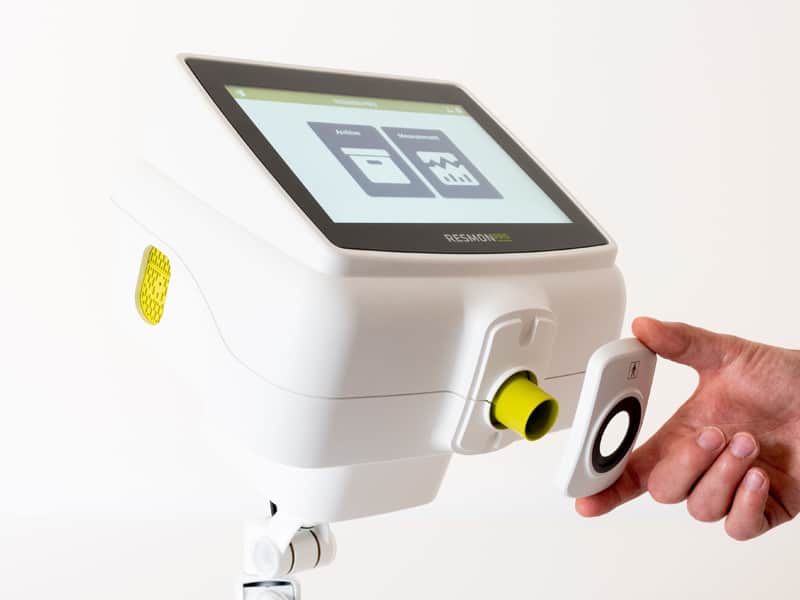
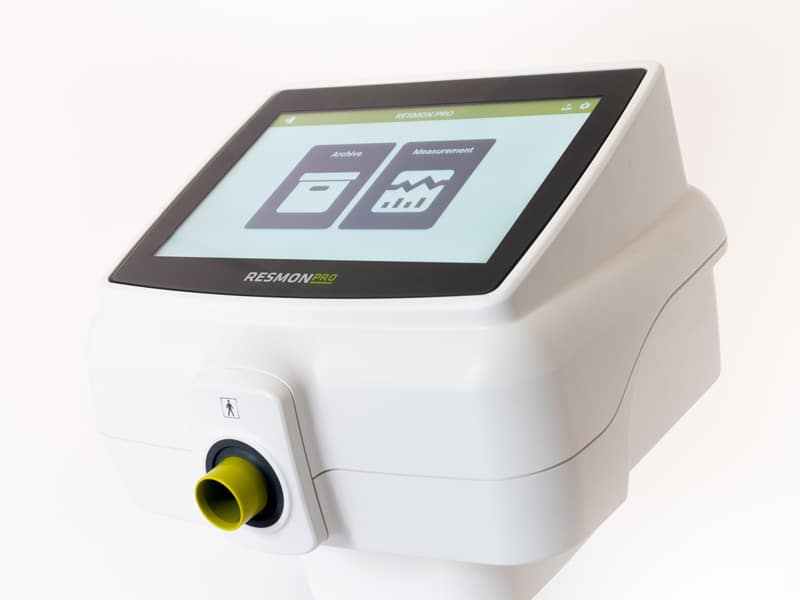
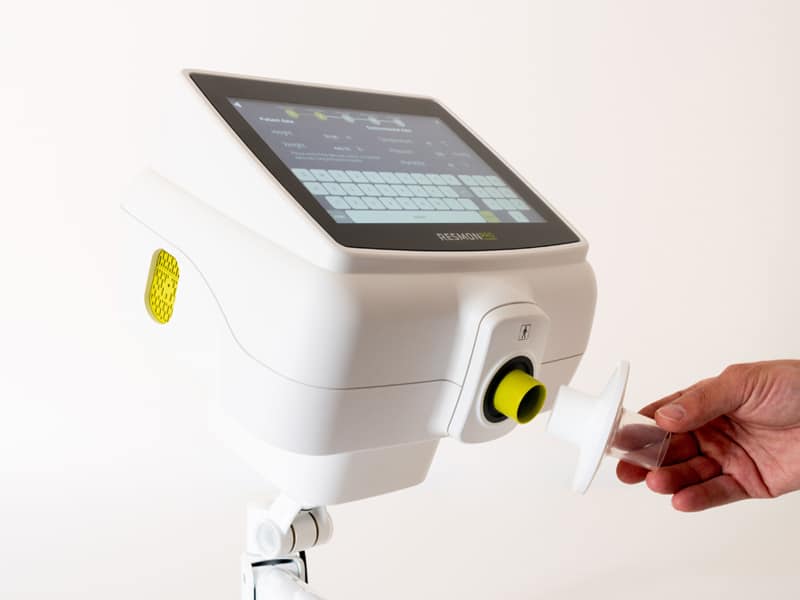
RESMON PRO FULL: a portable device with the features of a complete desktop system
With an innovative user interface developed with the support of cognitive experts and with new faster electronics, RESMON PRO FULL is a professional medical device based on FOT, developed for a complete, in-depth functional assessment of respiratory system. RESMON PRO FULL is a diagnostic complement to spirometry for pulmonologists, pediatricians, allergologists to easily assess in a single test:
- Presence of respiratory function impairment and its revesibility
- Localization of airway obstruction (central, small)
- Presence of tidal expiratory flow limitation (EFLt) using the patented index ∆Xrs obstruction
- Breathing pattern parameters (Ve,Vt, RR, Ti/Ttot and others)
- Slow vital capacity (SVC) and Inspiratory capacity (IC) – NOT AVAILABLE IN U.S.
Its standalone design and 10.1″ touchscreen make the units ideal for field testing.
Furthermore, thanks to its network capabilities and flexible, open data exporting features RESMON PRO is an optimal tool for research protocols and pharmacological trials.
RESMON PRO V3: the ONLY portable all-in-one device with the power of a stationary.
MAIN CERTIFICATIONS
- European CE Mark (MDD 93/42/EEC)
- US FDA 510k clearance
- Health Canada
- TGA (Australia)
- ISO 9001:2015 Quality Management Systems
- ISO 13485:2016 Medical Device Quality Management Systems
- MDSAP Medical Device Single Audit Program for Australia, Canada, Japan, US
CLINICAL AND RESEARCH USES
Pediatrics and adult pulmonary medicine, asthma, COPD, pulmonary and neurological rehabilitation, physiotherapy programs, effects of airways clearance evaluation, in-patient and out-patient uses, hospital and remote locations, Occupational medicine evaluation, sports medicine.
How does the RESMON PRO FULL WORKS?
The device uses oscillometry for measuring lung function during normal breathing. The assessment is based on the analysis of the respiratory system’s response to small pressure stimuli while breathing quietly for a few breaths in a mouthpiece connected to the device. A slow full inspiration followed by a slow full expiration executed at the end of the test allow the simultaneous assessment of lung volumes (IC and VC) giving a more comprehensive picture of the lung function.
What stimulating frequencies are available?
The following signals are available to measure within breath impedance:
- Sine waves: 5 Hz, 6 Hz, 8 Hz, 10 Hz
- Optimized pseudorandom noise (PSRN): 5-37 Hz
- Enhanced optimized multi-frequency: 5-11-19 Hz
What parameters can be measured?
- Inspiratory, expiratory and total Resistance for the detection of airways obstruction
- Inspiratory, expiratory and total Reactance for the detection of small airways impairment
- Breath by breath presence and degree of tidal expiratory flow limitation (EFLt) – PATENTED
- Spectral parameters like R5-19 and Fres
- Breathing pattern parameters, i.e. ventilation, respiratory rate, tidal volume, duty cycle, mean inspiratory and expiratory flow
- Vital capacity and Inspiratory capacity – NOT AVAILABLE IN U.S.